Reply To:
Name - Reply Comment
The National Medicines Regulatory Authority- the independent authority that regulates medicine and medical devices in the country- is continuously coming under severe criticism for failing to grant registrations to low cost brands of Rapid Antigen Test kits that have high efficacy and for issuing a registration waiver for a pharmaceutical drug that is still on a trial stage for COVID patients.
devices in the country- is continuously coming under severe criticism for failing to grant registrations to low cost brands of Rapid Antigen Test kits that have high efficacy and for issuing a registration waiver for a pharmaceutical drug that is still on a trial stage for COVID patients.
"The NMRA is also accused of the procedure they followed to offer the Rapid Antigen Test (RAT) tender to a party that has quoted a higher price"
This newspaper is in possession of a copy of the letter issued by the Director General / CEO of the National Medicines Regulatory Authority (NMRA) Dr. Kamal Jayasinghe granting a registration waiver to M/s George Steuart Health to import Ivermectin (12mg) one million tablets at a time; the drug is on a trial stage. This drug is manufactured by M/s Taj Pharmaceuticals Ltd, 214, Bake House, Bake House Lane, Fort, Mumbai, India.
The letter dated August 17, 2021, states further, ‘NMRA has granted approval to import and clear the above mentioned pharmaceutical item on the request made by Manager Regulatory Affairs by George Steuart Health (Pvt) Ltd of Sri Lanka.
‘This approval is granted in accordance with the powers vested on NMRA in Section 109 of the National Medicines Regulatory Act No: 5 of 2015 and shall be in force during the period stated in this letter unless earlier suspended or cancelled’.
"The tender was offered to the local agent of these two brands- George Steuart Health (Pvt) Ltd"
When contacted Dr. Kamal Jayasinghe to know why a registration waiver was given for a pharmaceutical drug that is still on a trial stage for COVID patients, he said he had no idea whether this drug is on a trial stage or not.
 “I have to check whether this drug is on a trial stage for COVID patients. There was a request from this company and there wasn’t any reason for us to reject it as this drug was available in the pharmacies before this importation. Those drugs are sold at a higher price. In order to bring down the prices we gave the applicant the waiver of registration,” Dr. Jayasinghe said.
“I have to check whether this drug is on a trial stage for COVID patients. There was a request from this company and there wasn’t any reason for us to reject it as this drug was available in the pharmacies before this importation. Those drugs are sold at a higher price. In order to bring down the prices we gave the applicant the waiver of registration,” Dr. Jayasinghe said.
A Senior Officer at NMRA speaking on terms of anonymity told the Daily Mirror that most of the officials at the NMRA were shocked to find out how an approval for waiver of registration could be granted when the drug in question is still on a trial stage for COVID patients.
George Steuart confirms receiving approval
George Steuart Health (Pvt) Ltd Executive Director Eran Ranasinghe confirmed they had obtained approval
|
Eran Ranasinghe |
for waiver of registration to the said pharmaceutical drug and added that the consignment had arrived in Sri Lanka from India on August 22 and got it cleared the next day (23).
“By now we have distributed the drug to pharmacies and each tablet will cost few rupees. There are counterfeit Ivermectin drugs in the market which are sold for exorbitant prices,” Ranasinghe said.
According to College of Medical Laboratory Sciences of Sri Lanka President Ravi Kumudesh questions have been raised as to why NMRA has granted approvals to George Steuart Health (Pvt) Ltd without a proper local validation. He said that the NMRA should take the responsibility if any catastrophe occurs.
The NMRA is also accused of the procedure they followed to offer the Rapid Antigen Test (RAT) tender to a party that has quoted a higher price.
"SPC Chairman Dr. Prasanna Gunasena appearing on TV last week said that only two brands of RAT kits had passed the sample validation when tenders were called last year"
When tenders were called in October 2020 by the State Pharmaceutical Corporation (SPC) to supply 100, 000 RAT kits, 36 companies submitted quotations, but only two products were selected. The tender was offered to the local agent of these two brands- George Steuart Health (Pvt) Ltd.
SPC Chairman Dr. Prasanna Gunasena appearing on TV last week said that only two brands of RAT kits had
|
Prof Channa Jayasumana |
passed the sample validation when tenders were called last year, but not the other brands.
Although Dr. Gunasena said so Ravi Kumudesh queried as to how SPC Chairman claims so when sample licenses were not issued by the NMRA to any of the tenderers on the advice of the College of Microbiology.
“How can the CPC Chairman say other than the qualified two samples all other 34 samples were rejected? None of the bidders were granted import licenses to get down samples and that was why the bidders were allowed to submit their bids without samples. Even the tender document specifically said that quotations can be submitted without samples. Although permission was sought from April 2020, by these suppliers for sample import licenses their requests were turned down. I challenge Dr. Gunasena to show proof that the other 34 bidders submitted samples for validation. The SPC knows well the blunder it created together with the NMRA to purchase these two test kits at a higher price and is now trying to cover up mounting criticism against them. They even had gone to the extent of lodging complaints against those who revealed these scams,” Kumudesh affirmed.
"Meanwhile, questions have been raised as to who granted sample import license to George Stuerts Health (Pvt) Ltd at a time when NMRA has banned issuing import licenses for RAT kits"
Several attempts made to contact Dr. Gunasena to obtain a comment proved futile. A text message was sent to Dr. Gunasena, but there wasn’t a response even at the time of going to press.
According to Kumudesh, it was surprising to note how NMRA claims that any product that has WHO approval doesn’t need to validate locally, although they refused WHO approved PCR test kits claiming NMRA cannot grant approval to any device even with the WHO approval unless it is locally validated.
Following the first tender being offered SPC General Manager Dinusha Dassanayake, when contacted, confirmed with the Daily Mirror that the first tender (No: DHS/RL/74WAC/20) was awarded to George Steuart Health (Pvt) Ltd. However Dassanayake declined to give further information regarding the selection procedure.
“I cannot divulge details as to on what basis the procurement was awarded to George Steuart Health,” Dassanayake said.
"According to Ranasinghe his company submitted samples of RAT kits to the NMRA in March 2020, for validation but the products were rejected"
When asked as to who is responsible in validating any drug or device that’s imported to the country Dassanayake said that it is up to the NMRA and not the SPC to check the validation process.
Meanwhile, questions have been raised as to who granted sample import license to George Stuerts Health (Pvt) Ltd at a time when NMRA has banned issuing import licenses for RAT kits even at the time the tender was floated in late October 2020.
|
Dr Kamal Jayasinghe |
According to Ranasinghe his company submitted samples of RAT kits to the NMRA in March 2020, for validation but the products were rejected.
“In March 2020, soon after the first COVID 19 outbreak, we were able to introduce sample test kits to NMRA, the IDH and other relevant institution to obtain the validation. But they were not happy with the sensitivity and specificity of the test kits and the products were rejected. We then informed the two manufacturers in Korea and they improved the product to suit our requirements. By April we once again produced these two brands for validation and the NMRA said that if we can get the WHO approval the local validation can be given,” Ranasinghe said.
Raising concerns, a cross section of the bidders who spoke to the Daily Mirror on conditions of anonymity, alleged that they were aware from the inception the government’s plan as the tender notice was not published in a national newspaper and instead on the SPC website; giving only six days, including a weekend, for the purpose of sending quotations.
"A Representative from WHO Sri Lanka told the Daily Mirror that these two brands have not undergone the process of full pre-qualification and they were approved only on an emergency listing."
“The SPC gave us short notice to submit our bids. The tender document stated that the entire quantity of 100000 kits for the qualitative detection of SARS Cov-2 antigen in human nasopharyngeal specimen with a swab and standard components have to be supplied immediately and the sensitivity and specificity of them should be very high. These test kits had to be delivered at the very earliest after winning the bid, considering its importance at this crucial time. We, the medical suppliers in March 2020, following the first COVID 19 outbreak requested the NMRA to issue sample licences to get down RAT test kits, but were turned down without a valid reason. If the winning bidder was granted a sample license, why couldn’t the NMRA give us the licence? When tenders were called in October we were asked to submit our quotations without samples. Had the NMRA issued us the sample licences on time, when requested in March, we would have submitted our samples for the local validation. We believe, the NMRA refused to issue sample import licences in March as they wanted to allow one company to import the devices. Although they claimed that WHO pre-qualified products can be used without any validation here, why didn’t they grant validation to PCR test kits that had WHO approval,” the sources claimed.
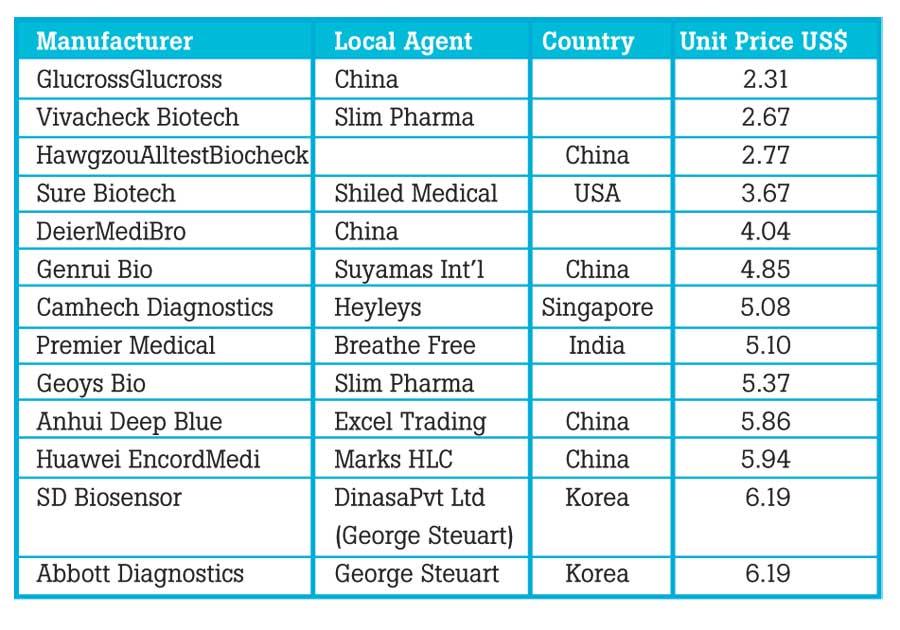
Be that as it may, according to copies of the SPC documents available with this newspaper there were eleven bidders that had submitted lower prices, than that of the price quoted by the winning bidder- George Stuerts Health (Pvt) Ltd. The successful bidder’s price for each unit was US$ 6.19 (SLR 1146.22) and the two selected brandsPanbio COVID-19 Ag Rapid Test (product code 41FK10) by manufactured by Abbott and STANDARD Q COVID-19 Ag, (product code 09COV30D), manufactured by SD Biosensor, Inc. are Korean products.
The products of the failed eleven bidders were from China, the USA, Singapore and India and the prices ranged from US $ 2.31 to US$ 5.94 per unit.
"According to copies of the SPC documents available with this newspaper there were eleven bidders that had submitted lower prices"
A senior Health Ministry official meanwhile raised concerns as to why the best products from USA and Singapore, which were amongst the low cost kits, were turned down by the SPC and NMRA.
|
Ravi Kumudesh |
“Other than the products from USA and Singapore there were products from China as well. State Minister of Pharmaceutical Production, Supply and Regulation Prof. Channa Jayasumana once said that they don’t want to purchase low quality Chinese RAT kits and that was why the tender was awarded to two WHO approved kits. If the State Minister says the Chinese brands are of low quality, then why did he remain silent when the Government placed orders for the Chinese vaccines? When he wanted to support one of his ‘Viyathmaga’ colleagues, he criticised the Chinese brands, but was supportive of getting down the Chinese vaccines which were given to the citizens of this country, but not to the government politicians, their families and friends,” Ravi Kumudesh alleged.
“Before any medicinal drug or device is imported the NMRA as the regulatory authority issues a sample import licence as it has the responsibility to check whether that device or drug, fulfills the local requirements for registration or use.” A Health Ministry source said.
However, the NMRA has followed these guidelines when issuing the sample licence to George Steuart Health (Pvt) Ltd of No: 7E, Post Masters Place, Off Templers Road, Mt. Lavinia on October 15. The licence has been issued subject to a local validation. But, due to unknown reasons the former had to revert the original decision on October 22, 2020 and issued the registration and the import licene to import the devices. Hence by November 3, 200, 000 test kits arrived Sri Lanka.
According to the Health Ministry Official all his attempts to get the NMRA stance to revert their October 15, 2020 directives to George Steuart Health failed as the Regulator was not willing to give an explanation.
"The products of the failed eleven bidders were from China, the USA, Singapore and India"
When questions were raised by the State Minister of Pharmaceutical Production, Supply and Regulation Prof. Channa Jayasumana and Director General/ CEO, NMRA Dr. Kamal Jayasinghe as to why George Steuart Health (Pvt) was allowed to import these medical devices without carrying out a local validation, both the State Minister and Director NMRA reiterated that any medicinal drug or device that has been certified by the WHO doesn’t need to be subjected to a validation process in Sri Lanka.
“NMRA Act No: 5 of 2015 stats that any medicinal drug or device that has been pre-qualified by the WHO need not be validated locally,” Dr. Jayasinghe said.
“There are two brands of Antigen Test kits that have been pre-qualified and pre-validated by the WHO. The
|
Dinusha Dassanayake |
accepted norm is, if the WHO recommends any drug or device we have no issue registering them without a sample validation. In this instance, George Stuart is the local agent for two brands that have WHO approval. That was why the NMRA gave the registration and the import licence to get down the test kits. These are for the private sector, but not for state hospitals. For the private sector, there is no need to follow any tender procedure,” Prof. Jayasumana said.
Although State Minister and Director General NMRA said that the two imported brands have been pre-qualified by the WHO and any device or drug that had been pre-qualified by them (WHO) does not need a local validation as per NMRA Act No: 5 of 2015, a Representative from WHO Sri Lanka told the Daily Mirror that these two brands have not undergone the process of full pre-qualification and they were approved only on an emergency listing.
Independent validation process
Responding to an e-mail sent by this newspaper, the Senior WHO Sri Lanka representative mentioned above stated that every country has the right to independently validate medical treatments or equipment, even though they may be WHO listed for emergency use, to fulfill the local requirements for registration or use.
The e-mail dated December 3, 2020 stated, ‘Given the urgent need posed by COVID-19, WHO HQ has issued guidance on the use of rapid antigen tests, and is currently evaluating some rapid Ag tests for Emergency Use Listing. WHO has granted emergency use listing to two tests – Panbio COVID-19 Ag Rapid Test (product code 41FK10) by manufactured by Abbott and STANDARD Q COVID-19 Ag, (product code 09COV30D), manufactured by SD Biosensor, Inc.
‘These tests have been approved on an emergency use listing and have not undergone the process of full pre-qualification. When WHO certifies a medicine or equipment, it makes this decision based on a review of reports submitted by the manufacturer which include data from validation studies conducted in several countries. This process increases the likelihood that the item can be safely used globally. Of course, every country has the right to independently validate medical treatments or equipment, even though they may be WHO listed for emergency use, to fulfill the local requirements for registration or use’.
NMRA Gave approval
When contacted soon after the tender was offered to George Steuart Health, Ranasinghe said that the NMRA, on October 22, 2020 had ordered them to get the local validation for these test kits, but was able to get the order reversed on the same day after a request was made to consider the WHO validation, considering the urgency of getting Covid tests done on BOI and EDB employees.
Refuting allegations leveled against George Steuart Health, Ranasinghe said that NMRA gave approval to import the RAT kits not based of any political connection, but on the safety and efficacy of the products.
“On September 22, 2000, we got the WHO approval for STANDARD Q COVID-19 manufactured by SD Biosensor and on October 2, 2020 for Panbio COVID-19 manufactured by Abbott,” Ranasinghe said.
When asked as to why the cost per unit is higher compared to other kits that have been submitted, Ranasinghe said that the two manufacturers have supplied 120 million kits to WHO at a rate of US $ 5 for their research purposes.
“In our tender document we stated our per unit price as Rs.1146.22 (US$ 6.19). We purchased a kit for US$ 5. After keeping a slim margin we quoted our price,” Ranasinghe added.
The Exchange Rate by the time the prices were quoted in November 2020, as per the Central Bank data was Rs 184.29 per one US$.
Considering the present Exchange Rate, when asked whether George Steuart Health would provide these kits at a loss now, as the dollar rate has gone up by a considering amount, Ranasinghe said they were able to get a price evaluation from SD Biosensor and Abbott Diagnostics
“When we informed them about the Exchange Rate, they reduced the test kit prices. That is why we still supply the same products for the same price without increasing as per the rising US Dollar rate,” he added.
Based on how the NMRA ordered George Steuart Health to get the local validation, but later reversed the order and issued the registration and import license, members of the College of Medical Laboratory Sciences of Sri Lanka and the medical experts challenged the Director of NMRA and the State Minister of Pharmaceutical Production, Supply and Regulation, explaining why such an order was given to George Steuart Health if there wasn’t a need to validate WHO pre-qualified drugs or devices locally.
“If NMRA now claims that WHO pre-qualified devices are not subjected to local validation as per their Act, then why did they ask George Steuart Health to get these two test kits validated locally? On whose directives did NMRA reverse the initial order and issued the license? Being an independent institution, why does the NMRA take orders from politicians? It is the same with the registration waiver granted to the same company to import one million Ivermectin tablets which is at the trial stage for COVID patients? Although the State Minister and the government officials are deceiving the general public, they cannot hoodwink those who are attached to the country’s health service,” Ravi Kumudesh, said.
Kumudesh further raised the question as to how George Steuart Health (Pvt) Ltd was granted a physically signed import licence at a time NMRA was issuing licenses and other related documents online due to the second wave of Covid -19 pandemic during October-November 2020.
“Why two different rules? Why did they give special privileges to George Steuart? Was it because of any orders received from the top?” Kumudesh queried.
According to Kumudesh, the sensitivity of a drug/device varies from one country to another.
“Since these two brands have been approved on an emergency use listing, but have not undergone the full pre-qualification process, we checked with the Food and Drug Administration of the USA and the Health Sciences Authority of Singapore. They have not given approval to these antigen test kits. How can the NMRA give approval to import such a device without checking its sensitivity to our people is questionable? Though the State Minister and the NMRA claim that these were imported for the private sector, they have forgotten that they too are used for our own people,” Kumudesh claimed.
According to him, there are nearly 100 manufacturers that produce antigen rapid test kits in the world, but by allowing only one party to import two brands, the ministry has deprived the country of the chance to buy a good product for a lower price after validation. “In order to get ready for a possible second outbreak, from April we were requesting from the Ministry to import antigen test kits, but until end October they did not take any action. The Ministry does not want to get advice from the members of the College of Medical Laboratory Services but only from Microbiologists,” Kumudesh said.