11 Nov 2020 - {{hitsCtrl.values.hits}}
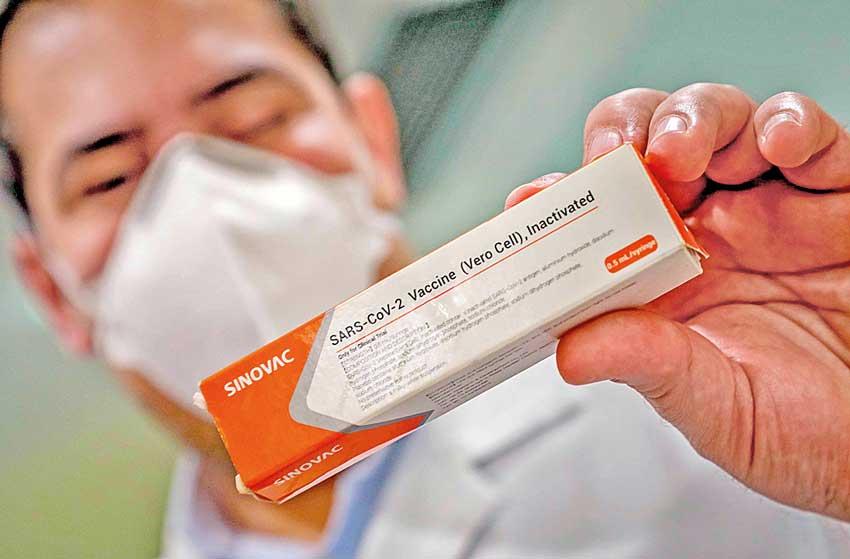
Brazil’s health regulator said it had suspended clinical trials of a Chinese-developed Covid-19 vaccine after an “adverse incident” involving a volunteer recipient, a blow for one of the most advanced vaccine candidates.
The setback for CoronaVac, developed by China’s Sinovac Biotech, came on Monday as US pharmaceutical giant Pfizer said its own vaccine candidate had shown 90 percent effectiveness --sending global markets soaring and raising hopes of an end to the pandemic.
Sinovac Biotech on Tuesday stood by its creation, saying: “We are confident in the safety of the vaccine”.
The Brazilian regulator Anvisa said it had “ruled to interrupt the clinical study of the CoronaVac vaccine after a serious adverse incident” involving a volunteer recipient on October 29. It said it could not give details on what happened because of privacy regulations, but that such incidents included death, potentially fatal side effects, serious disability, hospitalization, birth defects and other “clinically significant events.” Sinovac, however, said the incident was “not related to the vaccine”, adding it will “continue to communicate with Brazil on this matter.”
The public health center co-ordinating the trials of the vaccine in Brazil, the Butantan Institute, said it was “surprised” by Anvisa’s decision. CoraonaVac has been caught up in a messy political battle in Brazil, where its most visible backer has been Sao Paulo Governor Joao Doria, a top opponent of far-right President Jair Bolsonaro.
RIO, AFP JANEIRO,
Nov10, 2020
15 Nov 2024 3 hours ago
15 Nov 2024 4 hours ago
15 Nov 2024 5 hours ago
15 Nov 2024 5 hours ago
15 Nov 2024 6 hours ago